Type 1 Diabetes: When the Immune System Starts Misidentifying Itself
- Jason Lu

- Apr 25
- 4 min read

Introduction: The Problem Was Never Just Blood Glucose
If you’ve ever worked closely with Type 1 Diabetes (T1D), one realization comes quickly:
“Dysregulated blood glucose” is just the final outcome.
The real issue lies deeper — a systemic failure:
The immune system begins to attack the body itself.
This is why, despite over a century of insulin therapy, we’ve been able to manage T1D —but never truly solve it.
1. What is Type 1 Diabetes? (Type 1 Diabetes Overview)
At its core, Type 1 Diabetes is an autoimmune disease.
T cells within the immune system mistakenly recognize pancreatic β cells — the cells responsible for insulin production — as foreign, and progressively destroy them. This ultimately leads to:
Loss of insulin production
Hyperglycemia
Lifelong dependence on exogenous insulin
So the fundamental problem is not “insufficient insulin.”
It is that the cells responsible for producing insulin have been eliminated.
2. Causes of Type 1 Diabetes: Genetics × Environment × Immune Dysregulation
Genetics Is Risk, Not Destiny
T1D is strongly associated with HLA (human leukocyte antigen) genes.
However:
Concordance in identical twins is only about 30–50%
This suggests something critical:
Genetics sets the stage, but environmental triggers drive disease onset.
Environmental Factors: The Real Triggers
The current consensus is that T1D results from gene–environment interactions.
Key triggers include:
Viral Infections
Enteroviruses (e.g., Coxsackievirus B)
SARS-CoV-2
Potential mechanisms:
Direct infection of β cells
Molecular mimicry
Activation of immune responses
Environmental Stressors and Metabolic Pressure
BPA, PCBs (endocrine disruptors)
Air pollution (PM2.5)
Heavy metals
These contribute to:
Oxidative stress
Mitochondrial dysfunction
Immune dysregulation
Gut Microbiome
Emerging evidence suggests:
Reduced microbiome diversity
Compromised gut barrier (“leaky gut”)
Leading to:
Systemic inflammation
Breakdown of immune tolerance
Core Concept
T1D is not caused by a single factor.
It is the convergence of multiple processes:
β-cell stress + immune dysregulation + environmental triggers
3. Autoimmune Mechanism: Why Does the Immune System Attack β Cells?
From a mechanistic perspective, this is a multi-step amplification process.
β Cells Become “Visible” Under Stress
When β cells experience stress (oxidative stress, ER stress):
MHC class I expression increases
Antigen presentation is enhanced
Effectively, they become more visible to the immune system.
Emergence of Neoantigens
Under stress conditions, β cells generate:
Hybrid insulin peptides (HIPs)
These proteins:
Are not recognized as “self” by the immune system
Are treated as foreign antigens
Activation of T Cell–Mediated Attack
Key mechanisms include:
Molecular mimicry (virus → β cell cross-reactivity)
Bystander activation (inflammatory environment activates autoreactive T cells)
Treg dysfunction (failure of immune suppression mechanisms)
The outcome is progressive destruction:
The immune system continuously attacks β cells until they are nearly depleted.
4. Disease Progression: A Three-Stage Model
This staging is critical for understanding therapeutic strategies in T1D.
Stage 1
Presence of autoantibodies
Normoglycemia
Stage 2
Declining β-cell function
Dysglycemia
Stage 3
Clinical diabetes
Requires insulin therapy
This implies:
There is a therapeutic window before clinical onset.
5. Type 1 Diabetes Treatment
Insulin: Control Without Correction
Current standard of care includes:
Insulin injections
Continuous glucose monitoring (CGM)
Insulin pumps
However, these approaches:
Do not modify the underlying disease biology.
They compensate for loss — but do not restore function.
Immunotherapy: Beginning to Change Disease Trajectory
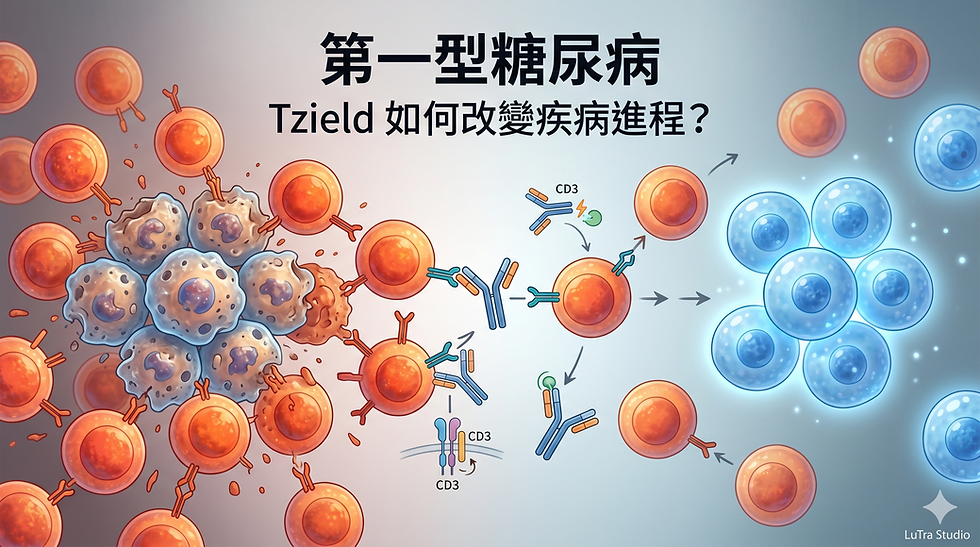
Tzield (teplizumab):
Anti-CD3 monoclonal antibody
Modulates T cell activity
Its significance lies in its mechanism:
Rather than broadly suppressing the immune system, it shifts T cells toward a regulated state.
Clinical Impact
Preservation of β-cell function (C-peptide)
Delay in disease progression
This represents the first clear evidence that:
T1D progression can be altered.
Cell Therapy: A Paradigm Shift
Islet Transplantation (Approved)
Lantidra:
Derived from donor islets
Improves glycemic control
Limitations include:
Requirement for immunosuppression
Limited donor availability
Stem Cell-Based Therapies (Emerging)
Recent studies show:
iPSC-derived β cells can enable insulin independence
However, major challenges remain:
Immune rejection
Long-term stability
6. Future Directions: Integration of Immunology, Cell Therapy, and Engineering
The future of Type 1 Diabetes treatment will likely not rely on a single solution, but rather on integration:
Immune Reprogramming
(Treg therapies, CAR-T, antibody-based modulation)
Cell Replacement
(Islet transplantation, stem cell-derived β cells)
Immune-Evasive Engineering
(Gene editing, encapsulation technologies)
This is no longer purely a medical problem.
It is a systems-level challenge involving biology, engineering, and manufacturing.
Translational Perspective: From Disease to Industry
From a biotech perspective, T1D represents a classic systems problem:
No single drug is sufficient
Platform-based approaches are required
Solutions span CMC, regulatory, and clinical strategy
This is why T1D remains a central focus of innovation in biotechnology.
At LuTra Studio, we focus on:
Evaluating biotech platforms and emerging technologies
Strategy development in RNA, cell therapy, and drug delivery
Translating science into actionable business insights
If you are thinking about:
How to advance a therapeutic platform into the clinic
Or how to evaluate the commercial potential of cell therapy
Feel free to connect.
Conclusion: This Is Not Just a Metabolic Disease
At its core, Type 1 Diabetes is defined by:
β-cell stress + immune dysregulation + environmental triggers
The key question moving forward is no longer:
“How do we control blood glucose?”
But rather:
“How do we stop the immune system from attacking itself?”
Final Thought
The future of Type 1 Diabetes will not be defined by better insulin.
It will be defined by our ability to redesign the relationship between the immune system and cellular function.
References
Tang et al., Environmental Mechanisms Influencing the Pathogenesis and Progression of Type 1 Diabetes, Int J Mol Sci, 2025
Ramos et al., Teplizumab and β-Cell Function in Newly Diagnosed Type 1 Diabetes, NEJM, 2023
Stabler & Russ, Regulatory approval of islet transplantation for treatment of type 1 diabetes, Mol Ther, 2023
Kumar et al., Stem cell therapy restores insulin independence in type 1 diabetes, World J Stem Cells, 2025
Rewers & Ludvigsson, Environmental risk factors for type 1 diabetes, Lancet, 2016




Comments