Teplizumab for Type 1 Diabetes: The First Time We Begin to Change Disease Progression
- Jason Lu

- 6 days ago
- 4 min read
Introduction: Treatment Is No Longer Just About Controlling Blood Glucose
For decades, the treatment of Type 1 Diabetes (T1D) has been relatively straightforward.
If the body cannot produce insulin, we replace it.
This approach works. It allows patients to manage the disease and maintain quality of life.
But if you look one layer deeper, a fundamental limitation becomes clear:
We have never truly changed the disease itself.
The problem is not just a lack of insulin.It is that the immune system continues to destroy β cells.
Which leads to a different question:
Can we change the progression of Type 1 Diabetes?
Tzield (teplizumab) emerged from this question.
1. What Is Tzield? (Teplizumab for Type 1 Diabetes overview)
Tzield (teplizumab) is an:
anti-CD3 monoclonal antibody
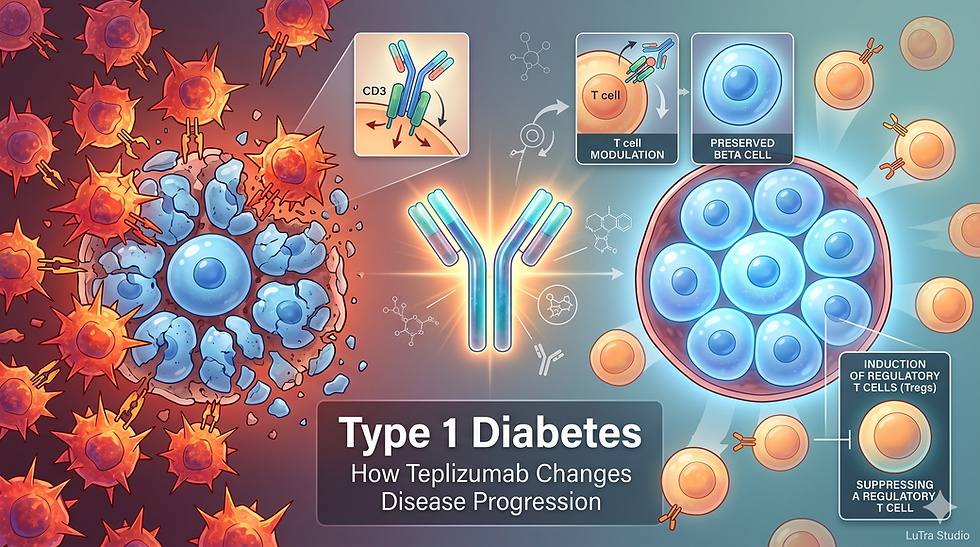
Unlike traditional therapies, it does not target β cells directly.Instead, it targets the immune system.
A simple way to think about it:
If Type 1 Diabetes is driven by:
the immune system attacking β cells
Then Tzield works by:
reducing the intensity of that attack
It does not shut down the immune system.It shifts its behavior.
2. Why This Represents a Fundamental Shift
At first glance, this may seem like a small difference.
In reality, it represents a major shift in how we think about treatment.
In the past:
Treatment = control blood glucose
Now:
Treatment = change the trajectory of Type 1 Diabetes
This is the core significance of teplizumab in Type 1 Diabetes:
For the first time, we are influencing the disease itself—not just its consequences.
3. Clinical Impact: What Tzield Actually Does
In clinical studies, teplizumab for Type 1 Diabetes does not eliminate the disease.
Instead, it slows it down.
In individuals at high risk (Stage 2):
It delays progression to Stage 3 by approximately 2–3 years
It preserves β cell function (measured by C-peptide)
This means:
The natural course of the disease can be altered.
This is the basis for its FDA approval.
4. Mechanism of Action: A Closer Look
CD3 and T Cell Activation
CD3 is part of the T cell receptor (TCR) complex.
When T cells are activated, signals are transmitted and amplified through CD3.
In Type 1 Diabetes, this signaling pathway contributes to the amplification of autoimmune attacks.
How Teplizumab Works
Teplizumab binds to CD3 and alters T cell behavior.
Current understanding suggests several effects:
Reduction of effector T cell activity
Induction of an exhaustion-like state in certain T cells
Enhancement of immune regulatory pathways
Because of this, teplizumab is better described as:
immune modulation rather than immunosuppression
5. Why Tzield Is Not a Cure for Type 1 Diabetes
This is the most important point to understand.
The immune system is not fully reset
Autoimmunity still exists—it is only partially modulated
β cells are not restored
There is no cell replacement, only preservation of remaining function
The effect is time-limited
Disease progression is delayed, but not stopped
In other words:
Tzield changes the speed, but not the endpoint.
6. Why Does Anti-CD3 Work?
Proposed mechanisms include:
Partial agonism
T cell exhaustion
Regulatory T cell modulation
However, the precise mechanism remains incompletely defined.
Why Timing Matters
Tzield is effective in Stage 2 but has limited impact in Stage 3.
This suggests:
Early intervention is critical.
Future Direction: Combination Therapy
It is unlikely that teplizumab alone will be sufficient.
Future strategies may involve combining it with:
Cell therapy
Antigen-specific tolerance approaches
Gene editing
7. Industry Perspective: What This Means for Biotech
The significance of teplizumab in Type 1 Diabetes goes beyond the drug itself.
It demonstrates a critical concept:
Immune modulation can alter disease progression.
This has implications for:
Drug development strategy
Clinical trial design
Investment decisions
More importantly, it opens a new space:
Treating disease before it fully manifests.
8. From a Single Drug to a Systems Strategy
If we step back, the challenge becomes clearer.
Type 1 Diabetes is not a single-problem disease.
It involves:
The immune system
Loss of β cells
Clinical and manufacturing translation
The real challenge is not finding a single effective therapy,but integrating multiple approaches into a viable system.
In practice, we often see technologies that make sense mechanistically,but fail at the clinical or translational level.
At LuTra Studio, we focus on these types of challenges, including:
Evaluating biotech platforms and emerging technologies
Strategy development in RNA, cell therapy, and drug delivery
Translating early research into clinical pathways
If you are working on:
Advancing a therapeutic platformor evaluating its long-term potential
The key question is not just whether it works mechanistically,
but whether it works as a system.
Conclusion: A Starting Point, Not the Answer
Tzield is the first therapy to demonstrate that:
Type 1 Diabetes progression can be delayed.
But it is not the final solution.
The fundamental question remains:
Can we stop the immune system from attacking β cells?
Final Thought
The future of Type 1 Diabetes will not be defined by better insulin.
It will be defined by whether we can redesign the immune system itself.
References
Ramos EL et al. Teplizumab and β-Cell Function in Type 1 Diabetes. NEJM, 2023
Herold KC et al. An Anti-CD3 Antibody, Teplizumab, in Relatives at Risk for Type 1 Diabetes. NEJM, 2019
Tang X et al. Environmental Mechanisms Influencing the Pathogenesis and Progression of Type 1 Diabetes. Int J Mol Sci, 2025
Sanofi Press Release, 2026



Comments