top of page
LuTra
Studio
LLC


Transforming Ideas into Impact.
Turning deep science, business insight, and career strategy into clear next steps.

Biotech & Biomedical Insights
Cutting-edge research, industry trends, and biotech career reflections — connecting science with society.


How I Self-Learned Molecular Biology Without a Biology Background
Introduction In the journey of cross-disciplinary learning, molecular biology is often the first real wall for anyone without a formal biology background . For those of us trained in engineering, the difficulty is rarely a lack of effort. Instead, it usually comes from three recurring challenges: Too many new terms and relationships appearing all at once Uncertainty about which concepts are essential and which can be temporarily set aside Learning ideas without knowing how th

Jason Lu
Jan 313 min read


Antibody–Drug Conjugates (ADCs): A Beginner-Friendly Deep Dive into Design, Linkers, Internalization, and Platform Strategy
This long-form article is written for readers without prior ADC expertise who want to understand why antibody–drug conjugates have become one of the most important oncology drug platforms of the 2020s. Starting from first principles, it gradually builds toward engineering trade-offs and industry strategy. Executive Summary Antibody–drug conjugates (ADCs) are designed to deliver extremely potent drugs directly into cancer cells using antibodies as targeting vehicles. ADC suc

Jason Lu
Jan 313 min read


Cancer Vaccines: From Immunology Theory to Personalized mRNA Therapies
Introduction: Why Cancer Vaccines Are Back in Focus For decades, the concept of a cancer vaccine occupied an uncertain space in oncology. Early therapeutic cancer vaccine efforts frequently failed to demonstrate meaningful clinical benefit, leading to skepticism about whether the immune system could be reliably trained to recognize and eliminate tumors (1). This perception has shifted dramatically in recent years. Advances in tumor genomics, neoantigen discovery, and RNA del

Jason Lu
Jan 255 min read


PEG-free LNP: How BioNTech Redefines mRNA Delivery Stability
Introduction In the world of mRNA therapeutics and lipid nanoparticles (LNPs), PEG-lipids have long been treated as a default design choice . They are used to: Provide steric shielding to prevent aggregation Extend circulation time Reduce nonspecific interactions However, as mRNA technologies move beyond one-time vaccines toward repeat-dose systemic therapies , PEG-associated risks—most notably anti-PEG antibodies and the accelerated blood clearance (ABC) phenomenon —are bec

Jason Lu
Jan 234 min read

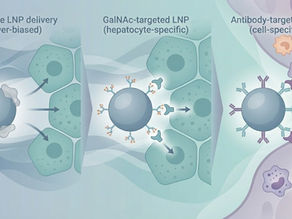
Targeted LNP Delivery: From GalNAc to Antibody-Guided Next-Generation Nucleic Acid Therapeutics
Introduction: The Next Bottleneck for LNPs Is Not Encapsulation — It’s Precision Lipid nanoparticles (LNPs) have proven their value by enabling the clinical success of mRNA vaccines and multiple nucleic acid therapeutics. However, as therapeutic targets expand beyond the liver to immune cells, the central nervous system, and solid tumors, non-specific biodistribution has emerged as a primary limitation of current LNP platforms . For many LNP-based systems, the key challenge i

Jason Lu
Jan 184 min read


GLP-1 Drug Price Reduction: Policy Shifts & Market Impact Analysis
White House × Lilly × Novo: A Deep Analysis of the GLP-1 Price-Reduction Agreement — Policy Shifts, Market Reactions, and Industry Impact Introduction GLP-1–based weight-loss drugs such as Ozempic, Wegovy, and Zepbound have become the most closely watched pharmaceutical products in recent years. Beyond their powerful impact on weight management, GLP-1 therapies are rapidly expanding into cardiovascular disease, obstructive sleep apnea (OSA), NASH, addiction, and other major c

Jason Lu
Nov 14, 20255 min read


The Role of Consulting in Biotech Success
In the fast-paced world of biotechnology, success hinges on more than just groundbreaking science. It demands strategic guidance, operational excellence, and market insight. Consulting plays a pivotal role in transforming innovative ideas into viable products and impactful solutions. I have witnessed firsthand how expert advice accelerates growth, mitigates risks, and opens doors to new opportunities. This post explores the critical role consulting plays in biotech success an

Jason Lu
Oct 16, 20253 min read


Mastering mRNA Synthesis: In Vitro Transcription, 5′ Capping, and Poly(A) Tail Engineering for Therapeutic mRNA
Introduction Messenger RNA (mRNA) has redefined what is possible in modern medicine. As a therapeutic platform, it enables rapid vaccine development, personalized cancer immunotherapy, and protein replacement strategies for rare diseases. However, behind every successful mRNA therapeutic lies a carefully engineered synthesis process. While clinical outcomes often dominate the conversation around mRNA therapeutics, the quality of mRNA synthesis ultimately determines translatio

Jason Lu
Jun 3, 20254 min read


The Rise of mRNA Therapeutics: From Genetic Messenger to a Transformative Medical Platform
Introduction Messenger RNA (mRNA), once a niche topic confined to molecular biology laboratories, has rapidly emerged as one of the most transformative technologies in modern medicine. While the COVID-19 pandemic brought mRNA vaccines into the global spotlight, their success was not an overnight miracle—it was the culmination of decades of foundational research, engineering breakthroughs, and persistent efforts to overcome the inherent fragility of RNA. Today, mRNA therapeuti

Jason Lu
May 18, 20255 min read


High-Throughput LNP Screening Using DNA and mRNA Barcoding Technologies
Introduction: The Delivery Problem in Nucleic Acid Therapeutics Nucleic acid therapeutics—such as mRNA vaccines, siRNA drugs, and CRISPR-based gene editors—have transformed modern medicine. However, the clinical success of these modalities depends not only on the therapeutic payload itself, but critically on how efficiently and selectively it can be delivered in vivo . Lipid nanoparticles (LNPs) have emerged as the gold-standard platform for systemic nucleic acid delivery. By

Jason Lu
May 12, 20255 min read


How Lipid Nanoparticles Enable Advanced Nucleic Acid Delivery: Engineering Principles of LNP Drug Delivery
Introduction Over the past decade, lipid nanoparticles (LNPs) have emerged as a cornerstone technology in nucleic acid drug delivery , enabling the clinical success of messenger RNA (mRNA), small interfering RNA (siRNA), and gene-editing modalities such as CRISPR-Cas9. Approved products like Onpattro® (patisiran) and the mRNA COVID-19 vaccines from Pfizer-BioNTech and Moderna have demonstrated that effective delivery—rather than payload design alone—is often the decisive fa

Jason Lu
Apr 28, 20254 min read


Job Interview Red Flags Series 1: Why “I Don’t Make Mistakes” Is a Major Warning Sign
Job Interview Red Flags: Why “I Don’t Make Mistakes” Is a Major Warning Sign (Interview Red Flags – Series #1) Introduction Many job candidates believe that presenting themselves as flawless is the safest interview strategy. However, from a hiring manager’s perspective, statements like “I don’t really make mistakes” are among the most common job interview red flags . This article examines why this response raises concern during job interviews, how interviewers interpret this

Jason Lu
Jan 22, 20254 min read


Design of Experiment (DoE): Principles, Advantages, and Impact on Scientific Research and Development
Introduction: When “More Experiments” Is No Longer the Answer In modern scientific research and engineering development, the central challenge is no longer whether to run experiments, but how to extract maximum insight from limited time, resources, and experimental capacity . Across drug development, bioprocess optimization, materials science, and platform R&D, researchers routinely face systems governed by multiple interacting variables. Traditional one-factor-at-a-time (OFA

Jason Lu
Sep 23, 20243 min read


Inside the Pharmaceutical Industry (III): How AI and Digital Transformation Are Redefining Drug Development
Introduction | AI in Pharmaceutical Drug Development The pharmaceutical industry is undergoing a profound shift as AI in pharmaceutical drug development and digital transformation reshape how modern medicines are discovered, tested, manufactured, and delivered. In Part I of this series, we explored the essential functions inside pharma companies. In Part II, we examined how drugs navigate the regulatory approval process, market access, and post-market surveillance. In this t

Jason Lu
Apr 1, 20244 min read


Inside the Pharmaceutical Industry (II): From Pharmaceutical Regulatory Approval Process to Market Access and Post-Market Surveillance
Exploring the Pharmaceutical Industry: An Overview of the Journey from Regulatory Approval to Market Access and Post-Market Surveillance. Introduction In Part I, we explored the key departments that drive the pharmaceutical industry — from R&D and CMC to clinical trials and business development. But even after a drug proves safe and effective in clinical trials, the journey is far from over. Before a treatment can reach patients, it must pass through one of the most complex p

Jason Lu
Mar 10, 20245 min read


Inside the Pharmaceutical Industry (I): Exploring Key Departments and Their Roles in Drug Development
The pharmaceutical industry is more than just lab coats and experiments — it’s a complex ecosystem where science, engineering, and business intersect. From R&D and manufacturing to clinical trials, regulatory affairs, and business development, each department plays a critical role in transforming a molecule into a life-changing medicine. Explore how these teams work together to bring innovation from the lab to the patient.

Jason Lu
Mar 4, 20246 min read


Biomedical Engineering Applications: How Engineers Transform Medicine — Lessons from Robert Langer
What engineers can do in biomedical field? Lessons from MIT and Robert Langer Engineers are trained to solve problems using science, mathematics, and systematic thinking. In our daily lives, civil, chemical, electrical, and software engineers continuously improve infrastructure, technology, and productivity. But what can engineers do in medicine? The answer lies in biomedical engineering applications —a field that translates engineering principles into real-world medical solu

Jason Lu
Nov 7, 20216 min read
bottom of page